Getting Ready for Advanced Therapy Medicinal Products (ATMPs) in Europe
Published July 2019

Recommendations for Timely Access to Transformative Therapies
In July 2019, ARM published a report on market access to ATMPs in key European countries. Designed to provide multi-stakeholder consensus on recommendations moving forward, the report is a result of the collaboration between ARM and European policy makers and experts, developers and manufacturers of ATMPs, patient organizations and other stakeholder groups. It provides a comprehensive review of the regulatory and market access framework across key European countries and identifies potential hurdles to the uptake of ATMPs in these areas. The report was funded by ARM.
ATMPs have the potential to offer a durable, life-changing therapeutic response, possibly with a single administration, for patients who may have few or no alternative treatment options. However, the complexity and relative novelty of these therapies present challenges to ensuring they reach those in need. While regulators have worked to adopt frameworks to ensure the appropriate, rapid approval of ATMPs in Europe, payers and health technology assessment bodies have not yet established the necessary specific mechanisms to ensure timely patient access to these therapies post-approval.
Getting Ready for Advanced Therapy Medicinal Products (ATMPs) in Europe
Report Recommendations
-
Recommendation 1: Better adapt Health Technology Assessment (HTA) frameworks to ATMPs
Better adapt Health Technology Assessment (HTA) frameworks to ATMPs by:
- Enhancing acceptability of validated surrogate endpoints to estimate long-term outcomes
- Conducting further research to improve methodology of indirect comparisons
- Supporting development, validation, and use of pan-European natural history datasets
- Leveraging scientific, clinical, and HTA expertise from centers of excellence
- Adopting changes in economic modeling, such as improving methods for extrapolation
-
Recommendation 2: Favor wider application of conditional reimbursement schemes
Conditional reimbursement schemes have the potential to mitigate uncertainty on duration of effect based on data available at time of regulatory approval. This approach is in use in several countries and a wider application in Europe for ATMPs is recommended.
-
Recommendation 3: Develop pan-European initiatives
Develop pan-European initiatives to create:
- Real-World-Evidence infrastructure: Real-World Evidence (RWE) development is instrumental in addressing uncertainties on long-term effect, safety, health-related quality of life, and use of healthcare resources. There is a need to develop RWE infrastructure and a common framework at the European level to support long-term evidence generation and procedures to enhance the quality of evidence collected specifically for ATMPs.
- New early-dialogue opportunities: There is a need for more opportunities for early-dialogue activities through additional EU and National funding. This would offer developers (and in particular SMEs) early insight on ways to address product specific uncertainties and how to mitigate them.
- Timely and effective access to cross-border healthcare for all EU patients: Despite existing legislation to facilitate cross-border healthcare in Europe, there are still barriers limiting access to ATMPs as they are most often delivered through centers of excellence which are not always present at the country or regional level. In particular, there is urgent need for measures to coordinate and fund access to cross-border ATMP treatment at European level.
-
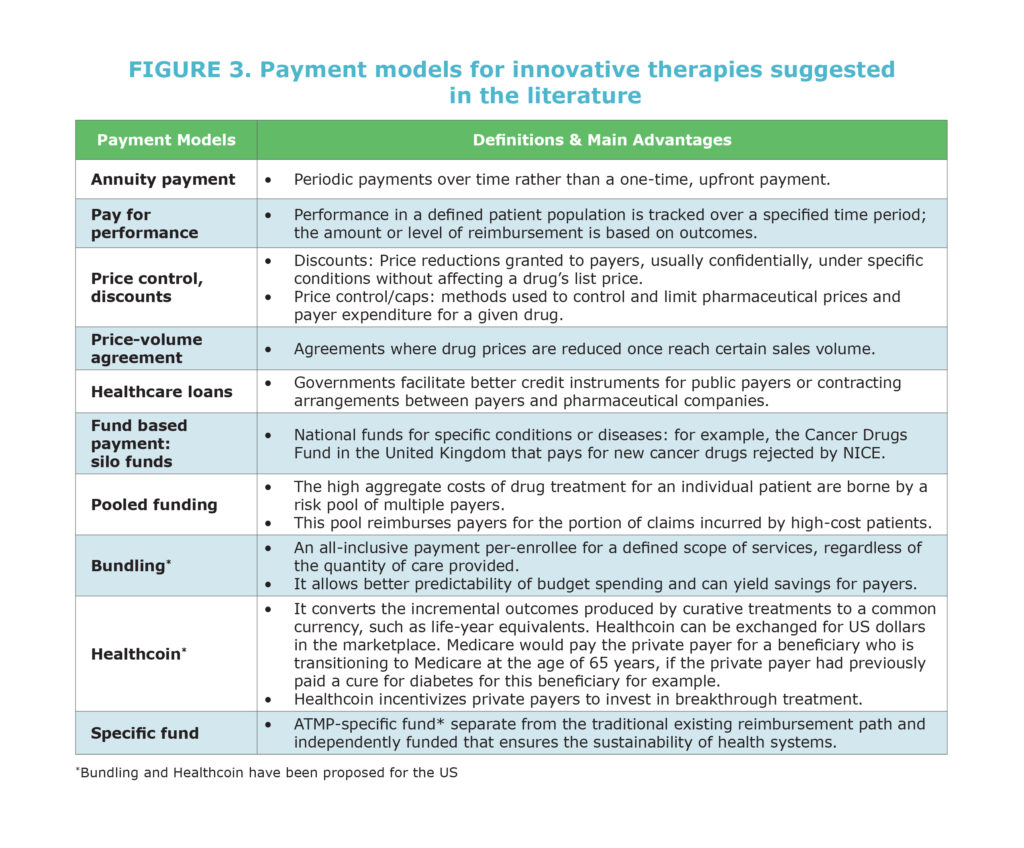
Recommendation 4: Favor wider application of innovative access and funding arrangements
New payment models are needed to ensure timely patient access to innovation while preserving sustainability of healthcare systems. Without the adoption of these new models, some transformative therapies may not reach patients in some or all European countries and some may be at risk of withdrawal from the market.
Innovative access and funding arrangements include:
- Pay-for-performance
- Annuity payments
- Special funds for transformative treatments
The Alliance for Regenerative Medicine hopes that continued dialogue and debate, supportive policy decisions, and a willingness among all stakeholders to create a fair and equitable environment for patient access to ATMPs will help overcome existing hurdles.
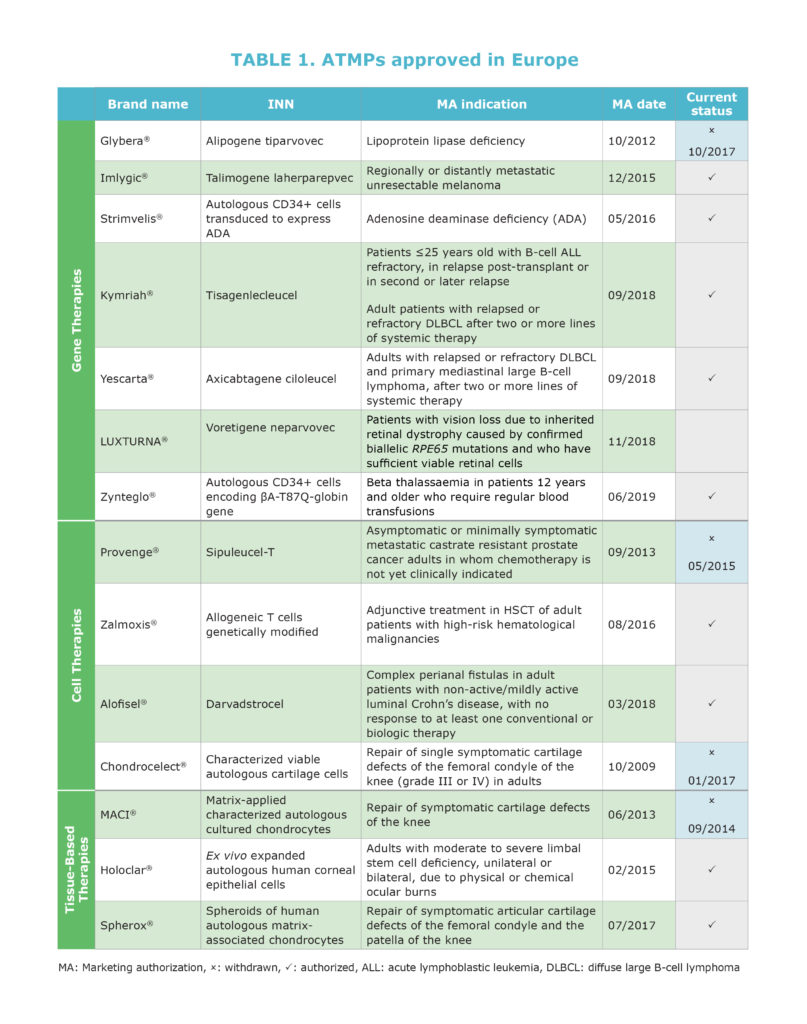
Marketed Products in the EU
Alofisel
TIGENIX
An allogeneic stem cell therapy to treat complex perianal fistulas in patients with Crohn’s disease.
Approved in: The European Union
Approved for: Complex perianal fistulas
Holoclar
CHIESI FARMACEUTICI
Holoclar is a cell therapy based on autologous cultures of limbal stem cells. It regenerates a functional corneal epithelium allowing recovery of visual acuity.
Approved in: The European Union
Approved for: Moderate to severe limbal stem cell deficiency due to ocular burns
Imlygic
AMGEN
Imlygic is a weakened form of Herpes Simplex Virus Type 1, which is commonly called the cold sore virus, indicated for the local treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma recurrent after initial surgery.
Approved in: The European Union and United States
Approved for: Unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma recurrent after initial surgery
Kymriah
Novartis
Kymriah is a CAR T-cell therapy product to treat R/R follicular lymphoma.
Approved in: The United States, the European Union, Canada, Japan, and Singapore.
Approved for: B-cell acute lymphoblastic leukemia, chronic lymphoid leukemia, diffuse large B-cell lymphoma, R/R large B-cell lymphoma, and R/R follicular lymphoma
Spherox (formerly Chrondosphere)
CO.DON AG
A product containing spheroids of human autologous chondrocytes for use in cartilage defects.
Approved in: The European Union
Approved for: Cartilage diseases
Strimvelis
ORCHARD THERAPEUTICS
Strimvelis is an ex-vivo stem cell gene therapy which uses retroviral vector encoding adenosine deaminase gene transfer into hematopoietic stem/progenitor cells.
Approved in: The European Union
Approved for: Adenosine deaminase severe combined immune deficiency
Zynteglo
bluebird bio
Zynteglo is an ex-vivo lentiviral based gene therapy used to provide a functional copy of the the β-globin gene into a patient’s hematopoietic (blood) stem cells.
Approved in: The United States (Withdrawn in the European Union in 2021)
Approved for: ß-thalassemia who require regular red blood cell (RBC) transfusions