
We are the leading international advocacy organization championing the benefits of engineered cell therapies and genetic medicines for patients, healthcare systems, and society.
Save the dates for the Cell and Gene Meeting on the Mesa!
The meeting, which runs from October 7-9, will take place at our new venue in Phoenix, AZ. Learn more!
Looking for updates on future ARM events? Sign up for our email list here!
Learn About Regenerative Medicine
Regenerative medicine includes gene therapies, cell therapies, and tissue-engineered products intended to augment, repair, replace, or regenerate organs, tissues, cells, genes, and metabolic processes in the body.
Regenerative medicine aims to alter the current practice of medicine by treating the root causes of disease and disorders.

FIND OUT MORE

Discover the Alliance
ARM takes the lead on the most important issues facing the regenerative medicine and advanced therapy sector today:
- Advocating for clear, predictable and harmonized regulatory and review pathways
- Enabling market access and value-based reimbursement policies
- Addressing industrialization and manufacturing hurdles
- Conducting key stakeholder outreach, communication and education
- Facilitating sustainable access to capital and identify sources of potential public funding.
Find Out More

Explore How ARM Leads the Way
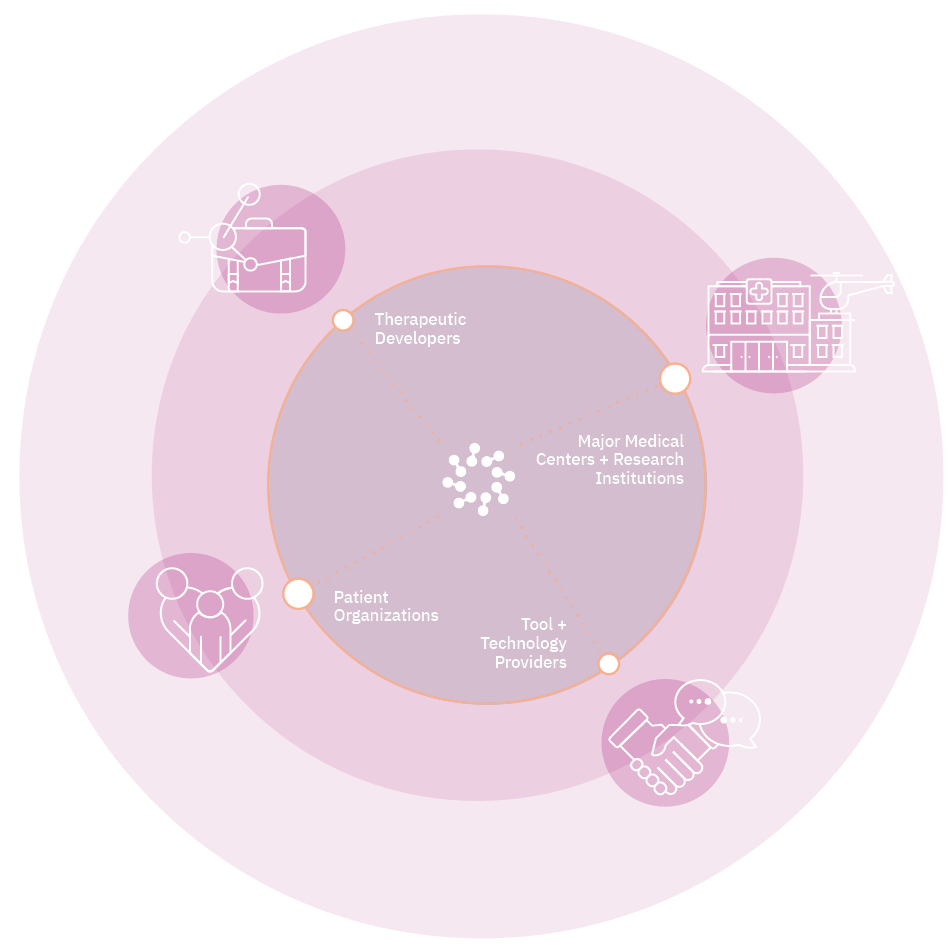
As the global voice of the sector, we represent more than 400 members across 25 countries, including emerging and established biotechnology companies, academic and medical research institutions, and patient organizations.
