Clinical Trials in Europe
Recent Trends in ATMP Development


The Alliance for Regenerative Medicine Outlines Recommendations for Increasing the Number of European-Based ATMP Clinical Trials
The report, which features both a quantitative analysis of clinical trial trends in Europe compared to other regions, as well as highlights from a survey of clinical-stage ARM members, provides recommendations for improving Europe’s competitiveness in clinical development compared to other global regions.
Key findings include:
- Though the number of ATMP therapeutic developers based in Europe is approximately half of that based in North America, the number of clinical trials that have been initiated in Europe during the 2014-2018 period is only about a third of that in North America.
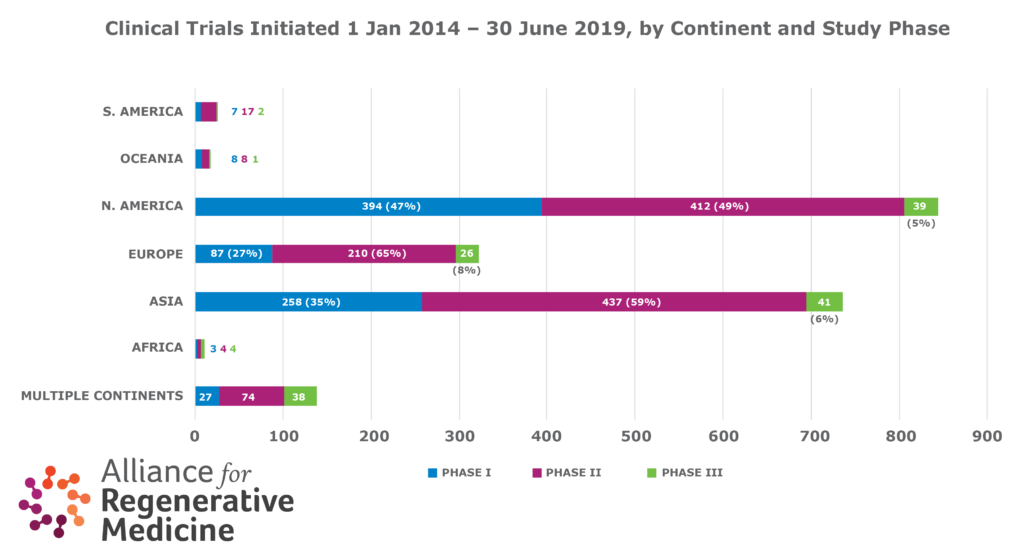
- During the same time period, the number of new clinical trials increased by 32% globally, 36% in North America, 28% in Asia, and less than 2% in Europe.
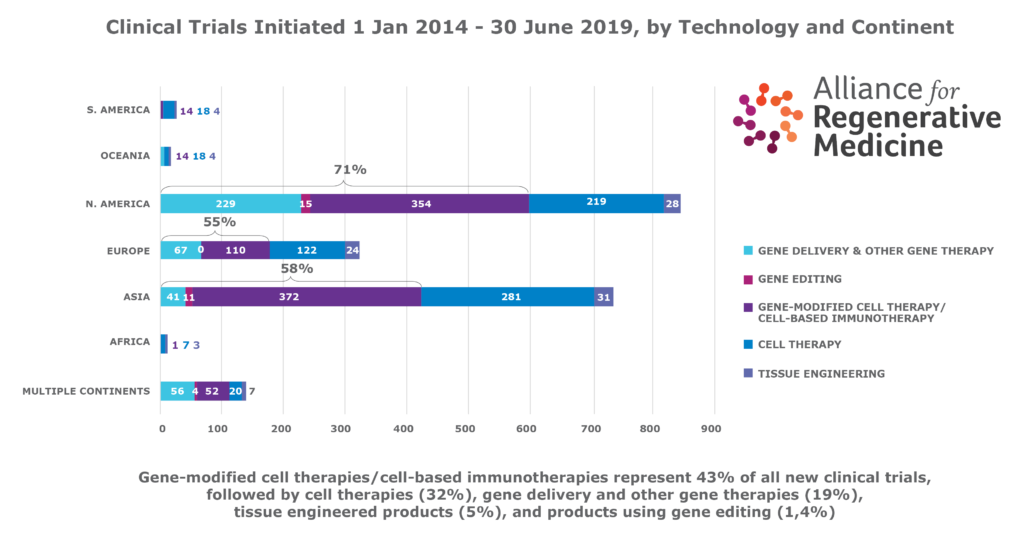
- There are proportionally more new gene therapy clinical trials (utilizing gene delivery, gene editing, and gene modified cell therapy technologies) in North America (71%) than in Europe (55%).
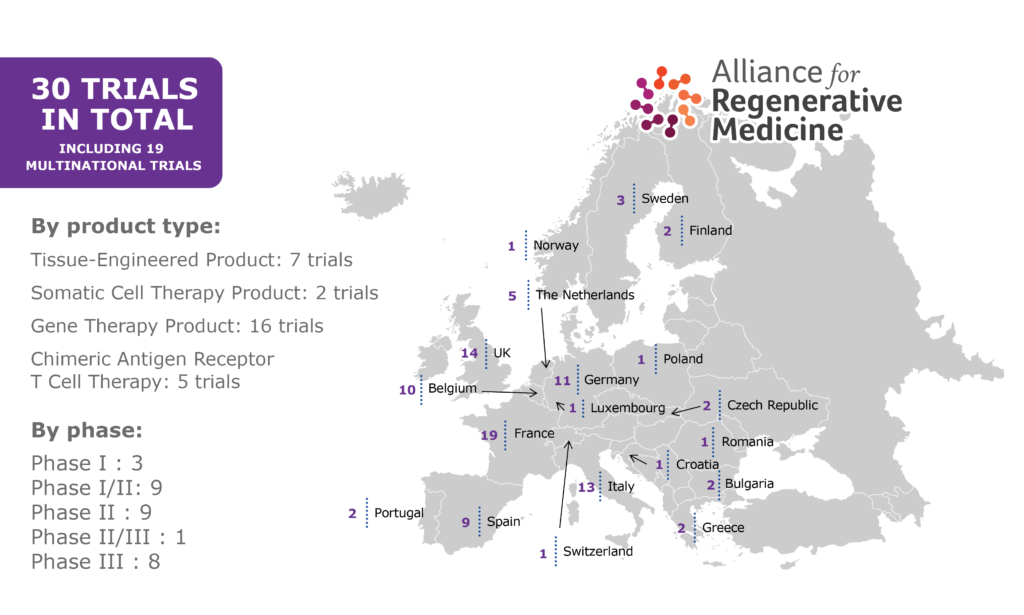
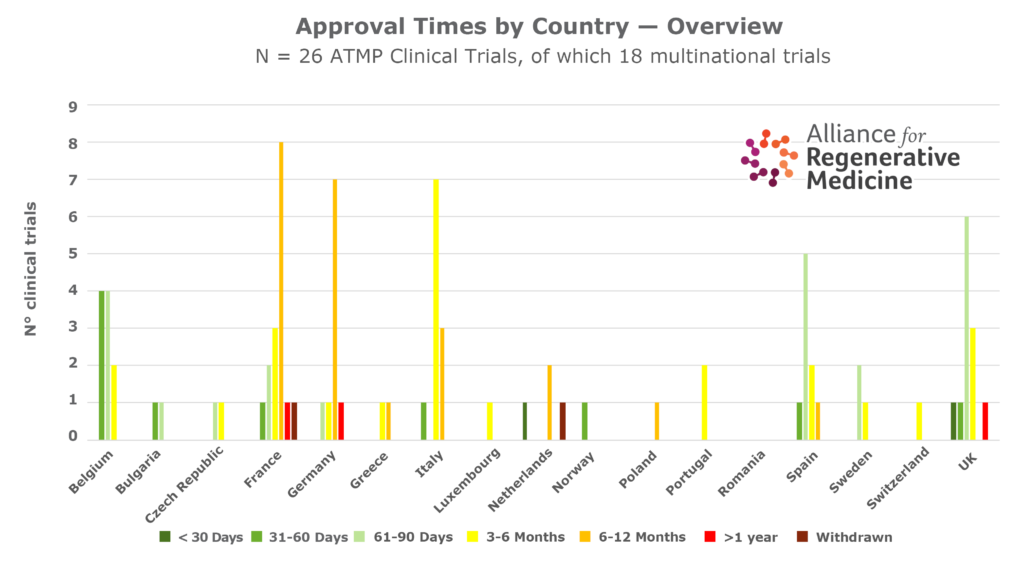
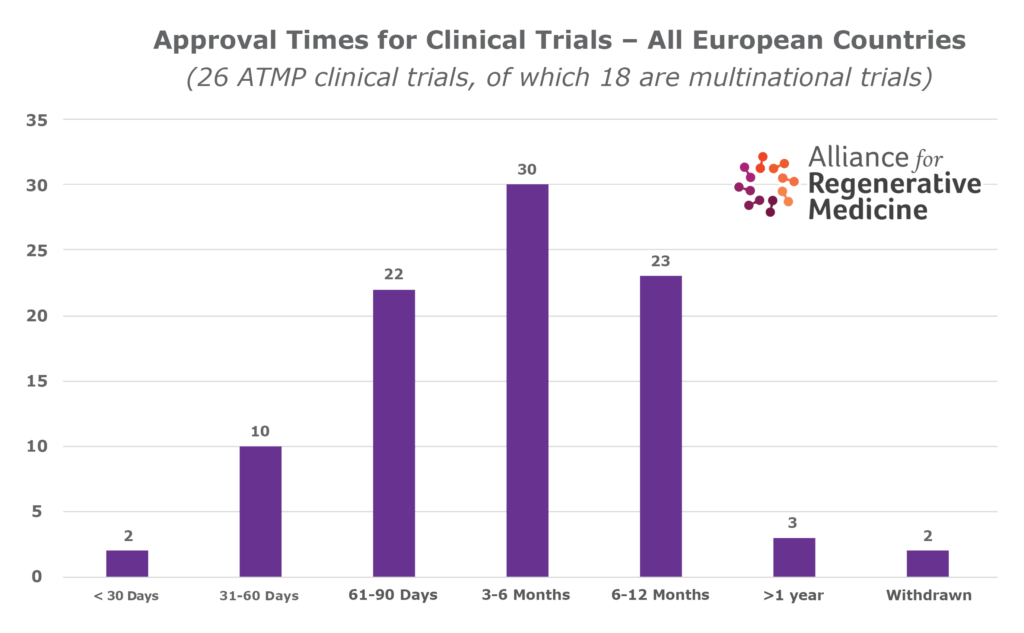
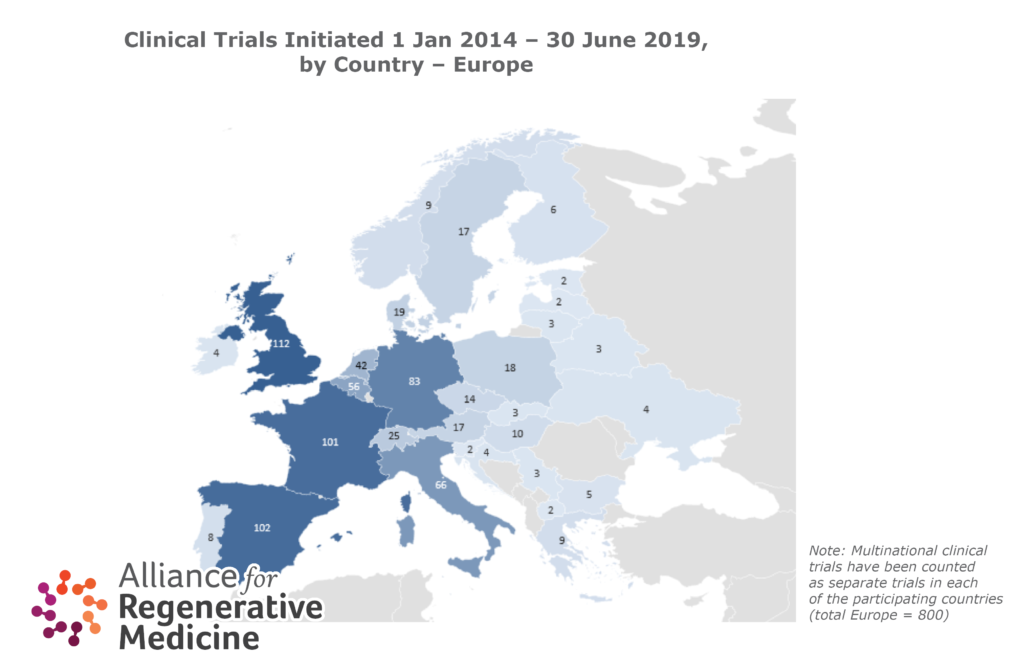
- There is considerable country-to-country variability in the number of clinical trials, speed of assessment, and time for approval of clinical trials in the different countries in Europe.
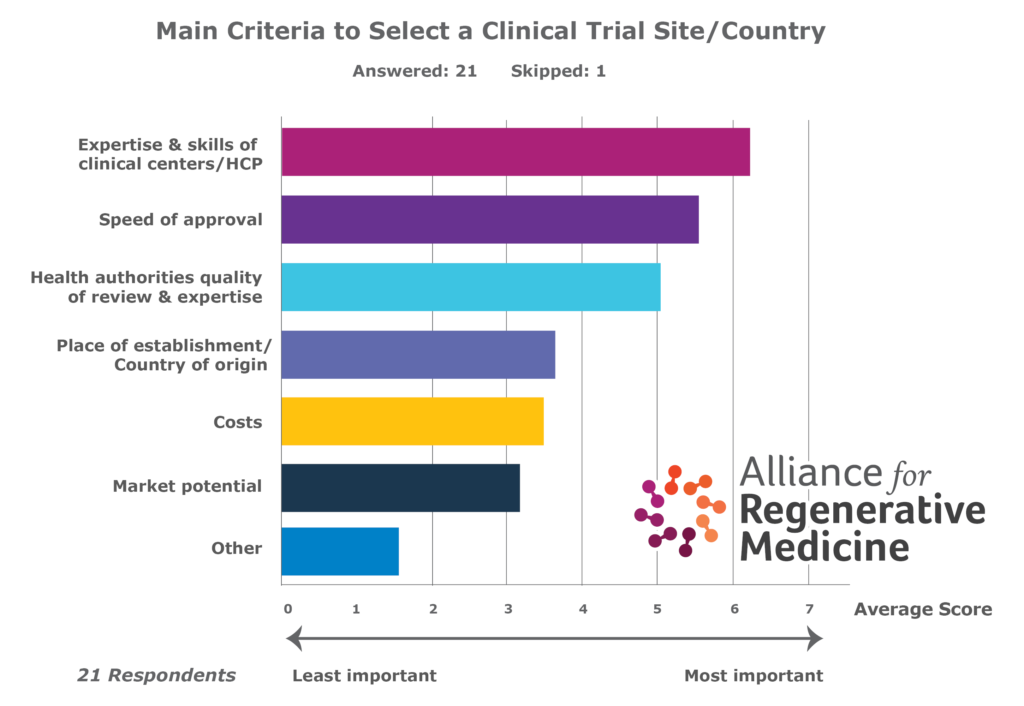
ARM recommends that European regulatory authorities improve timelines for the approval of clinical trials, streamline the process for the approval of multinational trials, implement a faster approval process for GMO review for gene therapies with a focus on consistency, and allocate sufficient resources and ensure an adequate level of expertise for the review of clinical trial applications for ATMPs. Regulators should view the implementation of the Clinical Trial Regulation (EU) No 536/2014 as an opportunity to streamline the review process and significantly improve approval timelines across Europe.