
UPCOMING EVENT
Cell & Gene Meeting on the Mesa
Location: 2400 E Missouri Ave, Phoenix, AZ 85016
Oct. 06, 2025
FIND OUT MORE
Location: 2400 E Missouri Ave, Phoenix, AZ 85016
Oct. 06, 2025
FIND OUT MOREARM, the International Society for Cell & Gene Therapy, and the American Society for Gene & Cell Therapy today called for a ten-year global moratorium on heritable human genome editing.
Adoptive cell therapy, the use of immune cells to help fight disease, is seeing rapid growth in the oncology industry. Its indications now span hematological and solid malignancies. ARM’s latest industry publication highlights the current state of this cutting-edge industry.
ARM's latest quarterly update on the cell and gene therapy sector is now available. Dive in for a recap of the notable sector clinical, regulatory, and investment trends from Q1 2025.
Dr. Peter Marks has been a visionary leader, advocate for rare disease patients, and champion of cell and gene therapy during his nine-year tenure as the Director of FDA’s Center for Biologics Evaluation and Research. He made many significant contributions to the field over the years and will be missed.
Regenerative medicine includes gene therapies, cell therapies, and tissue-engineered products intended to augment, repair, replace, or regenerate organs, tissues, cells, genes, and metabolic processes in the body.
Regenerative medicine aims to alter the current practice of medicine by treating the root causes of disease and disorders.


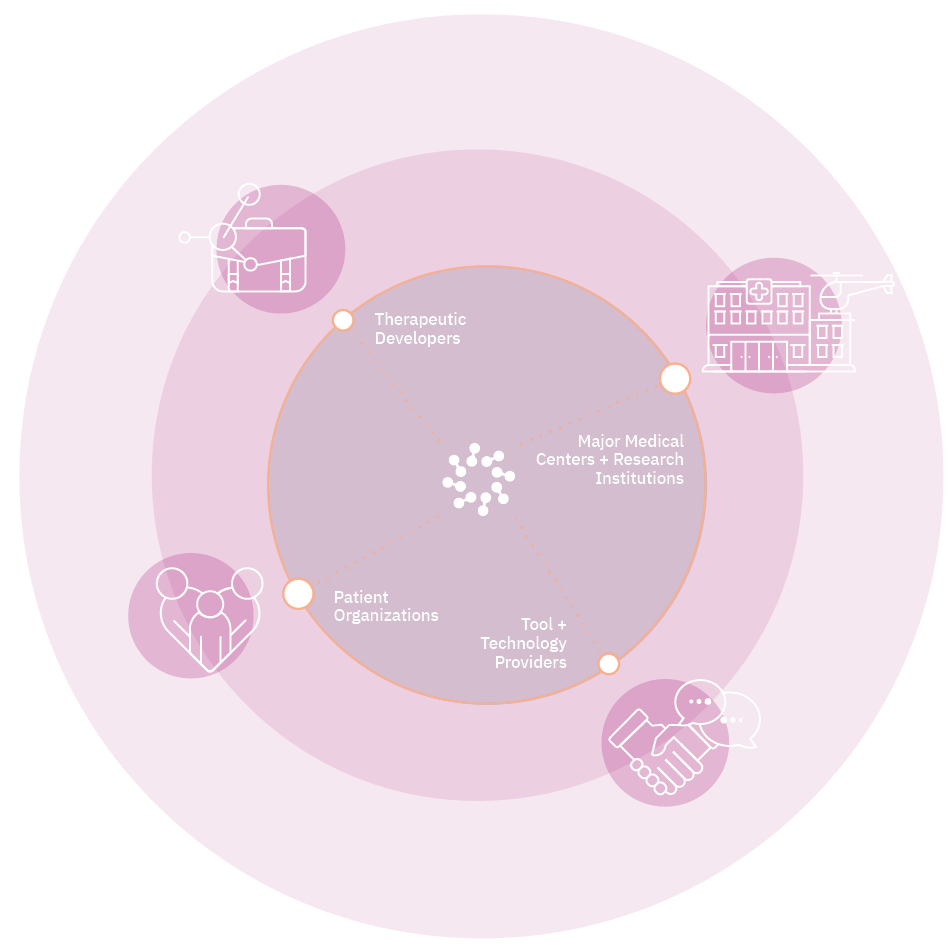
ARM is the global voice of the cell and gene therapy sector, representing more than 400 members across 25 countries, including emerging and established biotechnology companies, academic and medical research institutions, and patient organizations.

As a community, ARM builds the future of medicine by convening the sector, facilitating influential exchanges on policies and practices, and advancing the narrative with data and analysis.