Inherited Blood Disorders & Regenerative Medicine
This report provides industry specific statistics compiled from gene therapy, gene editing, cell therapy, and other regenerative medicine companies active in developing therapies for inherited blood disorders and provides context for the potential of regenerative medicine to improve upon the current standard of care.

As of March 2019:
There are 31+ companies worldwide working on regenerative medicine therapies for inherited blood disorders, including 18 clinical-stage companies.
There are currently 50+ products in active development with 38 clinical trials ongoing, including five in Phase III.
There are more than 20 million patients living with inherited blood disorders worldwide.
38+
Clinical trials underway
worldwide as of March 2019
PH. 1
7
PH. 1/2
24
PH. 2
2
PH. 3
5
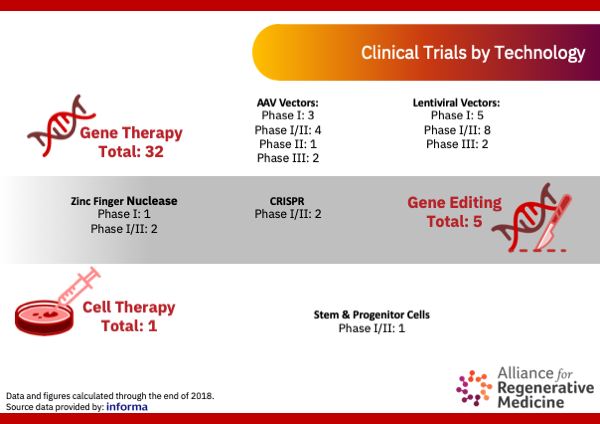
Clinical Trials by Technology Type
AAV Vector Gene Therapies
Lentiviral Vector Gene Therapies
Gene Editing Technologies
Cell Therapy
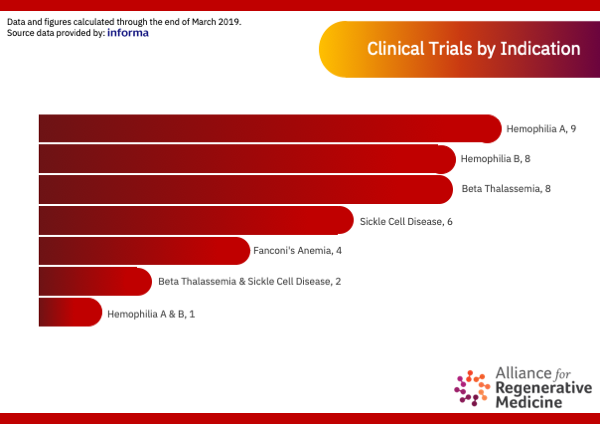
Clinical Trials by Indication
Companies Active in Developing Regenerative Medicines for Inherited Blood Disorders
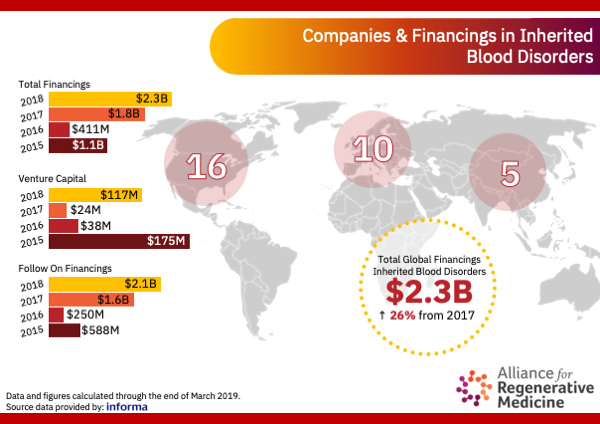
Financings
Global Financings for Regenerative Medicines for Inherited Blood Disorders
Total Global Financings
Inherited Blood Disorders
2018
$2.3 Billion
26%
Increase from 2017